Currently, vitamin D deficiency has been shown to cause a decrease in Hb concentration and an increase in RDW in children with TIDM, but no significant changes were observed in other blood indices. Some studies have shown that blood indices in children with TIDM did not change compared to control children.16,17 Except for Hb and RDW. In addition, other researchers pointed out that some blood indexes are different and change during the advanced stages of the disease.18The RDW is a standard complete blood count that measures changes in the size and volume of red blood cells and can also be used to identify the cause of anemia.7Incorporating RDW into the complete blood count evaluation facilitates diagnosis, especially of microcytic anemia.19Elevated RDW increases earlier than other red blood cell parameters and is therefore useful for early diagnosis of nutritional deficiencies such as iron and vitamin B12.20Increases in RDW occur not only in iron deficiency anemia but also in inflammatory anemia, with the former being higher.twenty oneHyperglycemia has various effects on red blood cells, including reduced deformability, shortened lifespan, and glycation of hemoglobin.twenty twoDM is associated with multiple microvascular complications leading to nephropathy, retinopathy, and neuropathy. Accumulating evidence suggests that alterations in red blood cells are both a cause and a consequence of the disorders associated with DM-related complications. Importantly, a significant proportion of DM patients develops anemia of varying degrees of unknown etiology, leading to increased morbidity. In chronic hyperglycemia, red blood cells exhibit morphological, enzymatic, and biophysical changes that predispose them to rapid phagocytic removal from the circulation.7RDW was higher in children with T1DM compared with normal children, whereas Hb levels were higher.20The present study showed a significant negative correlation between RDW and Hb in the vitamin D sufficient group. In children with vitamin D deficiency, RDW was negatively correlated with both Hb and serum iron, indicating a strong correlation between RDW and anemia, especially in vitamin D deficiency (Tables 1S and 2S).
Our study showed that serum iron, ferritin, TIBC, and transferrin were decreased in VD-deficient cases compared to controls with sufficient VD. Although no significant association was obtained between vitamin D and iron metabolism parameters in both groups of children, the results obtained indicated that children with VDD may be at higher risk of iron deficiency. Of note, some children in both study groups had low serum iron, but the proportion was higher in VD-deficient cases (7.6% of VD-deficient cases vs. 2.6% of controls with sufficient VD). Previous studies16 Although children with T1DM had normal range iron metabolism parameters and vitamin B12, this study did not show significant differences from healthy children in the control group, but ferritin and TIBC were lower in the T1DM group, which may indicate that the duration of the disease was not correlated with Hb, TIBC, serum iron, vitamin B12, and ferritin, respectively, indicating regular control and healthy nutritional status (Tables 1S and 2S).17.
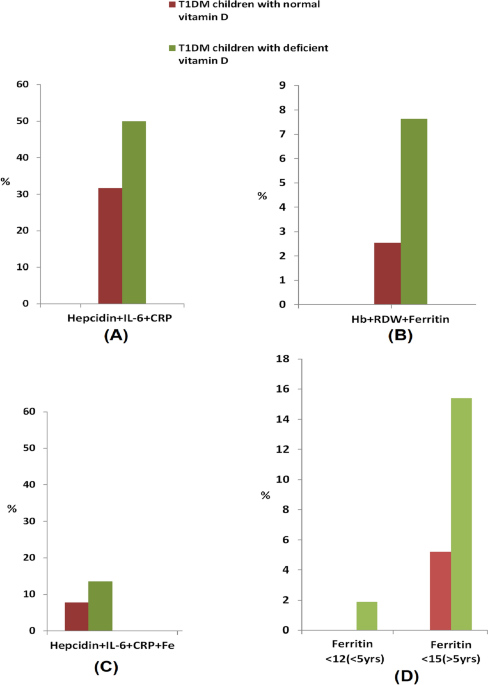
Our results suggested that the mean hepcidin levels were not significantly different between the two groups studied, but some children with insufficient VD had a higher rate of hepcidin elevation than those with sufficient VD, a behavior that has been observed previously.23,24,25Hepcidin is a systemic regulator of iron homeostasis. It is produced in response to iron stimuli and inflammatory conditions. Its role is to sequester iron in intracellular pools, reduce its gastrointestinal absorption, and reduce serum iron, a beneficial defense mechanism that masks iron use by bacteria.twenty oneIn this study, no significant difference in the mean hepcidin levels was found between the two groups of children with VD deficiency and those with sufficient VD, but the high incidence of VD deficiency in cases (67.3%) confirmed that VD deficiency suppresses hepcidin secretion. Currently, the median IL-6 levels do not show significant variation between the groups of children, whereas the median C-reactive protein levels were significantly elevated in the VD-deficient group of children with T1DM compared with matched values in the control group. VD has been shown to promote erythropoiesis by promoting the proliferation of erythroid progenitors and reducing inflammatory cytokines. Furthermore, VD may suppress hepcidin expression by reducing hepcidin-stimulating inflammatory cytokines and through direct transcriptional regulation of the hepcidin antimicrobial peptide (HAMP) gene. The reduction of proinflammatory cytokines and hepcidin may increase the bioavailability of iron for erythropoiesis and hemoglobin synthesis by restoring iron recycling, preventing iron sequestration in macrophages, and eliminating impaired iron absorption, thereby preventing anemia.26In this study, vitamin D deficiency increased the incidence of inflammation in vitamin D-deficient type 1 diabetic children compared with vitamin D-sufficient type 1 diabetic children, as indicated by elevated serum IL-6 and/or CRP. The elevated serum IL-6 and CRP reached 55.8% and 50.0%, respectively, in VD-deficient cases, compared with 39.5% and 31.6% of vitamin D-sufficient children. Furthermore, anemia of inflammation, as indicated by elevated hepcidin and the inflammatory markers IL-6 and/or CRP, was detected in VD-sufficient (31.6%) and VD-deficient (50.0%) children. Furthermore, a significant positive correlation was found between hepcidin and inflammatory markers in both the two groups of children in Table 1S and Table 2S, suggesting a strong association between inflammation and elevated hepcidin. This may also suggest a reduced state of chronic systemic inflammation in children with sufficient VD or ineffective erythropoiesis in T1DM children with insufficient VD.27.
It has been previously observed that diabetic factors may increase the risk of anemia, with anemia developing more quickly in diabetics than in patients with impaired renal function.28,29,30As can be seen from current research, vitamin D deficiency increases the incidence of anemia. People with vitamin D deficiency have been reported to have a higher prevalence and risk of anemia compared to those with sufficient vitamin intake.27The authors reported that the prevalence of vitamin D deficiency in type 1 diabetes patients was 48% compared with 36% in those with normal vitamin D status.
In conditions of VD deficiency, enhanced synthesis of hepcidin by hepatocytes or monocytes may increase intracellular and systemic hepcidin concentrations and decrease membrane expression of iron protein (Fp) in these cells. As a result, iron export is suppressed, leading to intracellular accumulation, increased cellular ferritin, and decreased systemic iron concentrations. Furthermore, decreased transcription of the HAMP gene may decrease intracellular and systemic hepcidin concentrations and simultaneously increase membrane expression of Fp. Enhanced iron export may decrease intracellular iron and ferritin, and increase systemic iron concentrations.twenty five.
It was previously reported that iron deficiency anemia can present as either absolute iron AID or FID.31,32The study concluded that vitamin D deficiency increases the incidence of both FID and AID in patients with type 1 diabetes. On the other hand, the incidence of AID was increased in cases of vitamin D deficiency depending on the age of the children studied. However, some other studies have shown iron deficiency in patients with type 1 diabetes without distinguishing between AID and FID.33,34However, previous studies have explored the possibility that there may be two types of type 1 diabetes.35,36A high incidence of AID and FID has also been previously shown (5.7% and 47%, respectively).36.