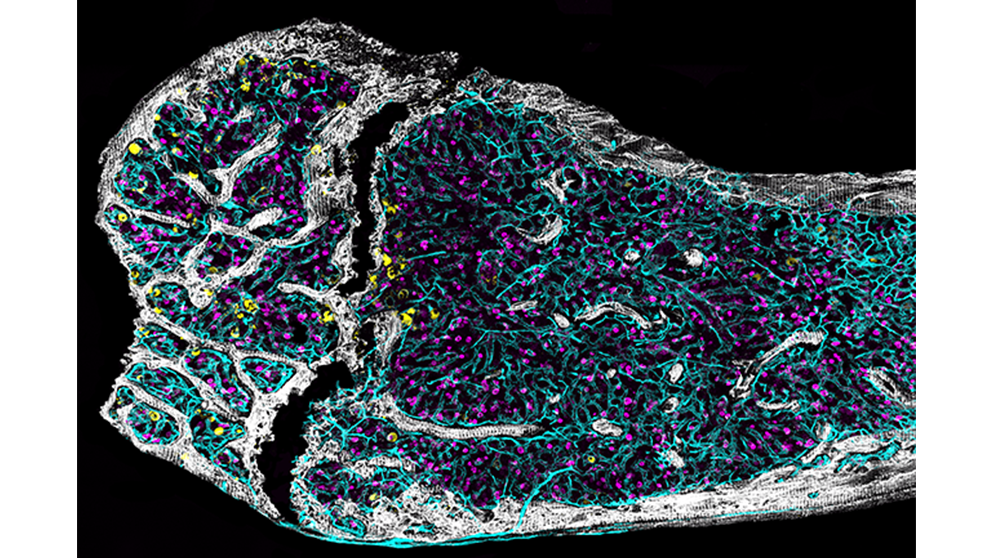
image: Immunofluorescence images show detailed morphology and cellular composition of femurs from middle-aged mice. credit: Jennifer Trowbridge
Human bone marrow is the gelatinous substance found inside your bones and is one of our body’s invisible powerhouses, silently churning out around 500 billion new blood cells every day. This process is driven by hematopoietic stem cells, which generate the various blood cells in your body and also self-renew to keep the process running smoothly.
Hematopoietic stem cells and aging
Hematopoietic stem cells are not immune to aging, and their function declines with age, leading to increased risk of serious diseases such as blood cancer. Although the risk of developing age-related diseases varies from person to person, little is known about whether hematopoietic stem cell aging also differs from person to person.
“If you put a group of 50-year-olds in a room, some of them will have completely gray hair, some will have a mix of gray and white hair, and a few will have no gray hair at all,” says Jennifer Trowbridge, Dattels Family Chair and professor at the Jackson Laboratory. “Logically, we would expect to see similar differences in hematopoietic stem cell function, but no one has studied it directly until now.”
Research into hematopoietic stem cells has been limited in part because they are so rare: they are usually all studied together. bloodThe researchers studied hematopoietic stem cells at the single-cell level in nine genetically identical middle-aged mice to examine how subtle changes in the bone marrow microenvironment lead to aging of hematopoietic stem cells in individual mice.
What they found
The researchers found that hematopoietic stem cells in the bone marrow of individual mice aged differently, even though the mice were the same age. Furthermore, the researchers were able to predict the function of hematopoietic stem cells based on the activity of two growth factors present in the human body.
Kitl and Igf1 growth factors are produced by mesenchymal stromal cells (MSCs) that surround stem cells in the bone marrow, and profiling of the RNA transcriptomes of individual mouse MSCs revealed that a decrease in these two growth factors correlated with an age-associated molecular program in hematopoietic stem cells.
“We found that the amount of growth factors produced directly correlated with the decline in stem cell function, with the changes being more pronounced in hematopoietic stem cells than in other cells in the bone marrow,” Trowbridge said. “This is really a snapshot of the aging process at a cellular level.”
In genetically diverse humans with diverse lifestyles, the variability in hematopoietic stem cell aging may be even greater than that observed in the carefully controlled animal model used in this study.
The researchers say that while the study did not investigate whether stem cell senescence directly causes adverse health effects, the changes likely influence a range of outcomes in both mice and humans.