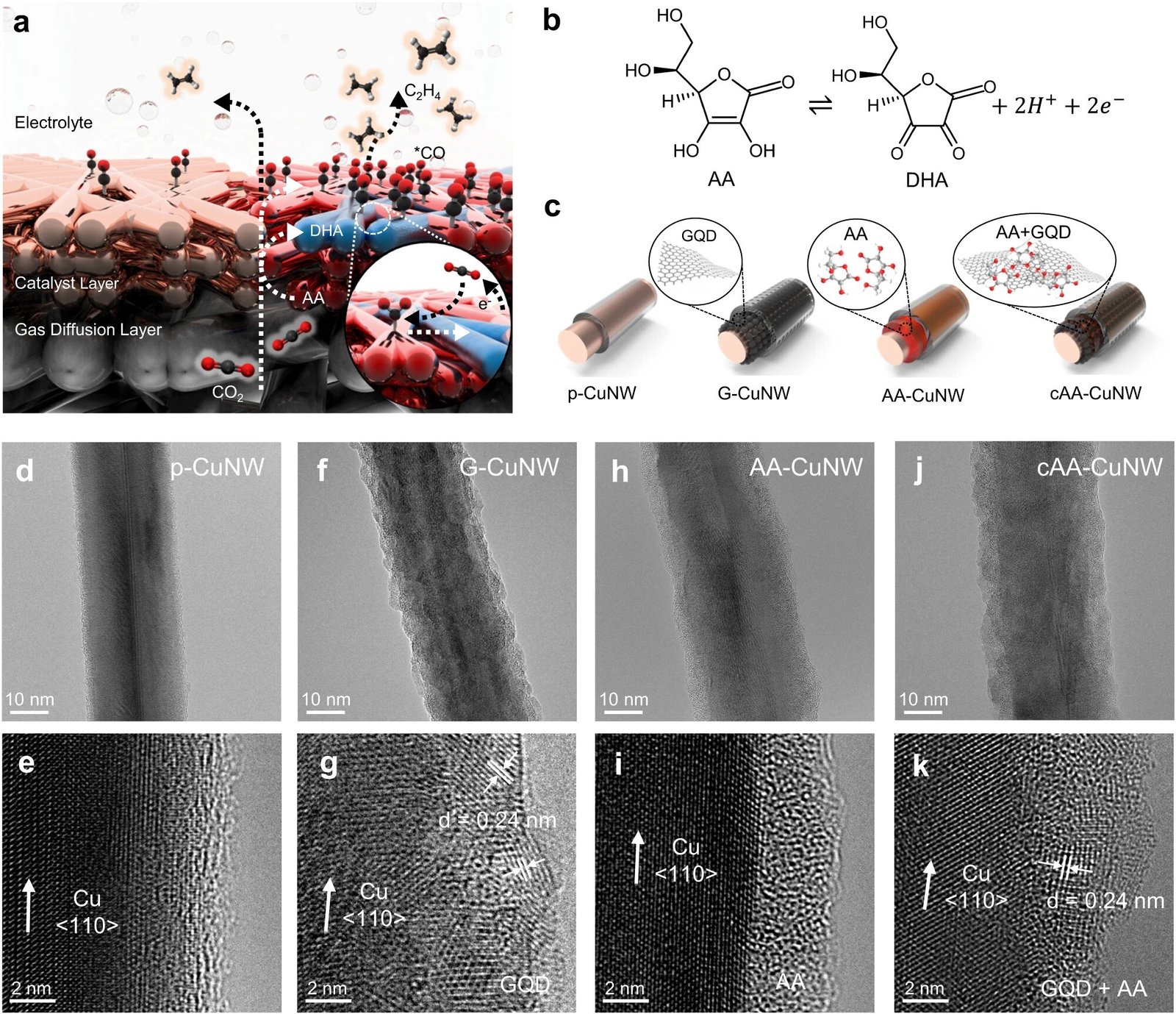
C.O.2 Capture strategy and surface structure of AA-enhanced CuNWs. be Schematic diagram of enhanced CO2-to-*CO conversion and *CO dimerization in cAA-CuNWs with high percentage C2HFour production. b Redox of AA and DHA to CO2 capture. c Schematic illustration of surface modification of CuNWs with GQDs, AA, and nanoconfined AA on GQDs. During the fabrication of GDE, an ionomer is coated on the outer surface of his CuNWs. TEM (top) and HR-TEM (bottom) images of (d, e) p-CuNW, (debt, g) G-CuNW, (ah, I) AA-CuNW, and (j, k) cAA-CuNW. credit: nature communications (2024). DOI: 10.1038/s41467-023-44586-0
× close
C.O.2 Capture strategy and surface structure of AA-enhanced CuNWs. be Schematic diagram of enhanced CO2-to-*CO conversion and *CO dimerization in cAA-CuNWs with high percentage C2HFour production. b Redox of AA and DHA to CO2 capture. c Schematic illustration of surface modification of CuNWs with GQDs, AA, and nanoconfined AA on GQDs. During the fabrication of GDE, an ionomer is coated on the outer surface of his CuNWs. TEM (top) and HR-TEM (bottom) images of (d, e) p-CuNW, (debt, g) G-CuNW, (ah, I) AA-CuNW, and (j, k) cAA-CuNW. credit: nature communications (2024). DOI: 10.1038/s41467-023-44586-0
A joint research team has developed a new electrochemical catalyst to accelerate the conversion from carbon dioxide (CO).2) to ethylene (C2HFour).
Through joint research by Professors Dae-hyun Nam and Professor Yung-Lee of DGIST’s Department of Energy Science and Engineering, and Professor Seo-in Back of Sogang University’s Department of Chemical and Biomolecular Engineering, the research team devised a technology. Incorporation of vitamin C into a heterogeneous carbon dioxide reduction catalyst significantly enhances ethylene production.
This is based on the observation that the presence of carbon dioxide in the air affects vitamin C levels in fruit.
The technology to electrochemically reduce carbon dioxide is attracting attention as a fundamental technology for “environmentally friendly energy.” This process aims to reduce carbon dioxide levels in the atmosphere while producing cleaner energy sources for the future. However, existing electrochemical catalysts face challenges in achieving consistent catalytic performance under high current density conditions.
This restriction prevents the production of carbon monoxide, an essential intermediate product that plays a key role in ethylene conversion. Rather, these catalysts tend to induce hydrogen evolution reactions rather than carbon dioxide reduction reactions.
Therefore, in order to seamlessly reduce carbon dioxide, an electrochemical catalyst must stably generate a carbon monoxide intermediate at high current density and promote dimerization in which two carbon monoxide intermediates combine. is important.
Therefore, a research team led by Professor Nam of DGIST developed a method to integrate the redox reaction of vitamin C with the electrochemical reduction of carbon dioxide, based on the phenomenon that the vitamin C content of fruits decreases in high-concentration environments. devised. of carbon dioxide.
The research team synthesized vitamin C using graphene quantum dots, and created “copper nanowires that enhance vitamin C” by combining the synthesized material with copper. This approach helped stabilize vitamin C through the nanoconfinement effect of graphene quantum dots and enabled redox reversibility.
In addition, the redox reaction of vitamin C constantly supplies electrons and protons to carbon dioxide, facilitating the dimerization process and producing carbon monoxide intermediate product. As a result, the newly developed catalyst showed a 2.9-fold improvement in ethylene production compared to the conventional copper nanowire catalyst.
Additionally, through real-time Raman spectroscopy and computer simulations, the researchers found that vitamin C trapped in graphene optimized the integration of carbon monoxide intermediates and copper catalysts. The researchers also identified the catalyst’s working principle by examining the electron and proton transfer that drives the carbon dioxide reduction reaction based on strong hydrogen bonds.
Professor Nam of DGIST said, “This research created an electrochemical catalyst that is capable of large-scale ethylene production through the reduction of carbon dioxide, and revealed a new reaction mechanism. It is expected that this will play an important role in achieving carbon neutrality.” Converts the main cause of global warming into high-value compounds. ”
The work will be published in a magazine nature communications.
For more information:
Jongyoun Kim et al., Vitamin C-induced CO2 Recovery enables high-speed ethylene production in CO2 electrical reduction, nature communications (2024). DOI: 10.1038/s41467-023-44586-0
Magazine information:
nature communications
Provided by: DGIST (Daegu Gyeongbuk Institute of Science and Technology)