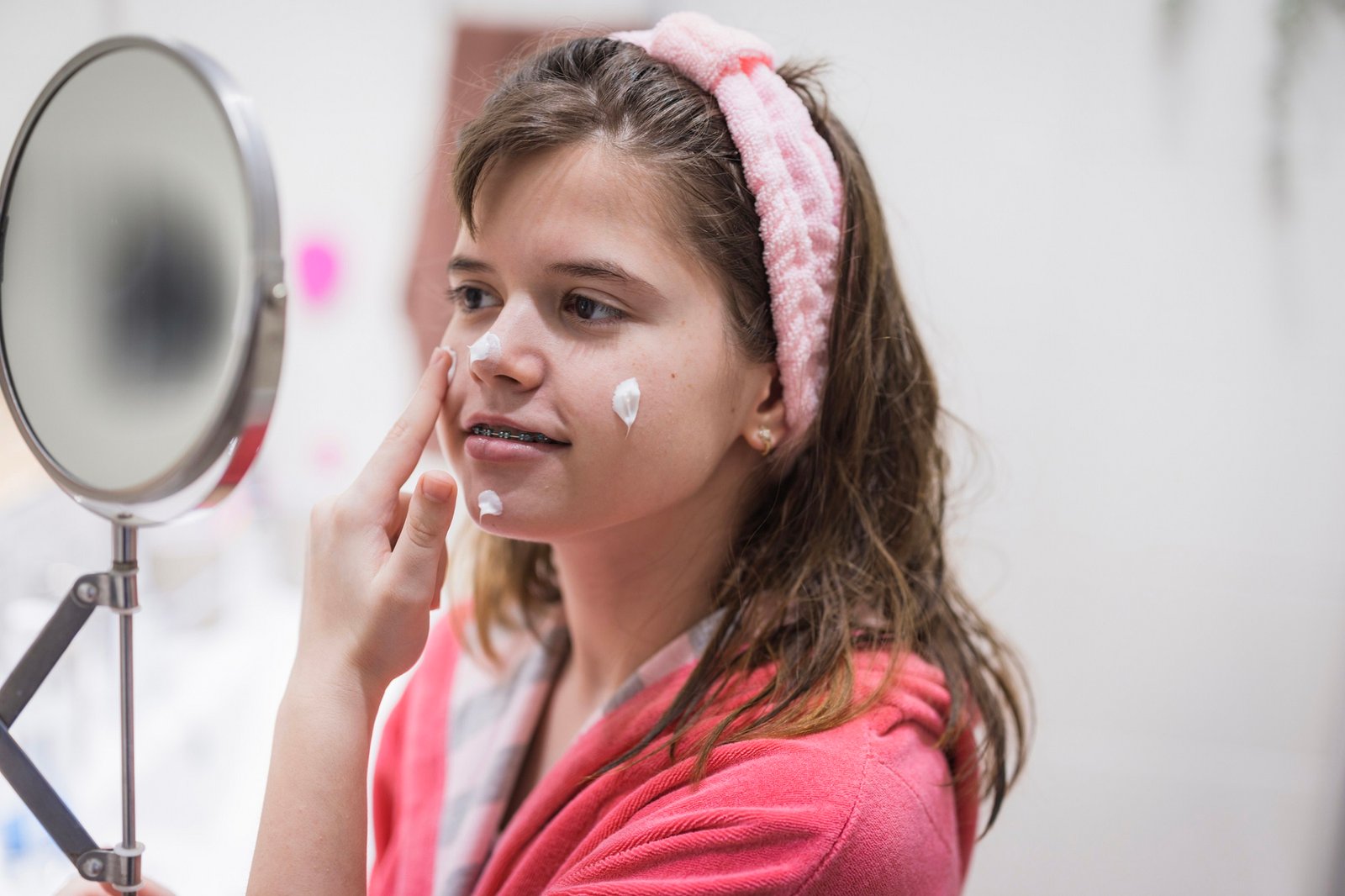As reported by multiple outlets in recent months, including the New York Post, CNN, and BBC, there has been growing concern about the inappropriate use of anti-aging skin care products by teen and pre-teen consumers. In response to this “skin care boom,” California Democrat Congressman Alex Lee introduced a bill in February this year aimed at banning the sale of “over-the-counter skin care products or cosmetics advertised to address skin aging that contain any of the following intentionally added ingredients: Vitamin A and its derivatives (including but not limited to retinoids, retinol), and/or alpha hydroxy acids (including but not limited to glycolic acid, ascorbic acid (vitamin C), and citric acid).”
California Assembly Bill 2491 was initially passed by the Environmental Safety and Hazardous Substances Committee in April but was not heard by the state Assembly last month. While the bill was being considered by the state Assembly, it drew strong reactions from the national cosmetics and personal care products industry, including the Personal Care Products Council (PCPC), which issued a statement saying the bill “further than addressing real issues, it creates regulations that are so complex they are nearly impossible to comply with and enforce” and “threatens to over-regulate products that are essential to safe and healthy skin care.”
Given that the bill did not move forward, the question remains whether other states will follow suit by drafting their own (amended) versions of AB 2941. To understand the outlook for future regulatory action in this area, CosmeticsDesign reached out to the PCPC as well as industry attorneys Kelly Bonner, associate attorney at Duane Morris LLP, and Anne Begley, partner at Wiley Rein LLP and chair of the firm’s Food and Drug practice. When asked their views on the issue, here is what each of them said:
Personal Care Products Council
“While we share Rep. Lee’s concerns about the popularity of preteens and teens using anti-aging products on social media, California’s bill, AB 2491, however well-intentioned, raises significant compliance issues, including by requiring cashiers to verify product claims and customers’ ages at checkout,” PCPC said.
“This bill threatens to restrict essential products like sunscreens, moisturizers and cleansers that are safe for mature skin. PCPC and our member companies want to ensure parents, guardians and teens and pre-teens have access to reliable information and increased resources about skin care for pre-teens so they can make educated choices about skin care products.”
Ann Begley, Partner at Wiley Lane LLP
“If the bill had passed, it would have required businesses doing business in California to take ‘reasonable’ steps to ensure that purchasers of teen-restricted products are not under the age of 13,” Begley said. “‘Reasonable steps’ would have been to post a prominent notice next to the physical product or in the product’s online description, or to take steps to verify the purchaser’s age. Failure to take such steps would have resulted in civil penalties of up to $10,000 per day per violation.”
“The cosmetics industry agreed that the social media-driven trend of encouraging teenagers to use products aimed at older audiences is concerning, but opposed California Assembly Bill 2491, arguing that the regulation is too complex and broad in scope to be both complied with and enforced. There are countless skin care products that contain these ingredients, many of which are not labeled or intended as ‘anti-aging,’ and therefore targeted cosmetic sellers will have difficulty complying with the bill’s requirements.
“For example, if the ID requirement were adopted as proposed, retailers would have had to show many customers ‘ID’ to avoid potentially being fined $10,000 per violation. To reduce the risk of violations, retailers could choose not to stock products containing restricted ingredients unless they were clearly labeled so that retail staff could easily identify products that are subject to ‘tween restrictions.
“Alternatively, retailers may have chosen to limit stocking of restricted products, even when clearly labelled, to further manage compliance risks. In response, manufacturers may have had to make costly labelling changes or even more costly formulation changes to replace the pre-teen restricted ingredients.”
“Further, the California Assembly Appropriations Committee determined that the bill would impose an unknown but significant amount of operational cost pressures on the Department of Justice’s (DOJ) General Fund. With the General Fund facing a structural deficit of tens of billions of dollars over the next few years, the Committee clearly had concerns about moving forward with this bill.”
“While California Assembly Bill 2491 did not make it to debate, it’s possible that new legislation will be introduced during the next legislative session, and that lawmakers in other states may introduce similar legislation if the social media trend continues. Realistically, educating parents and children is likely the best approach. The cosmetics industry could aid in this effort by using its social media presence to combat the current trend.”
“However, if a legislative solution is pursued, it should take a more targeted approach and aim to ban the promotion, rather than the sale, of the products. In this case, retailers, manufacturers and paid influencers could be prohibited from advertising or promoting skin care products containing these ingredients to people under the age of 13, unless there is scientific evidence that the concentrations used in the products are safe for use in this age group.”
“Notably, evidence of prohibited promotional activity could include monetary payments or other material rewards directed at influencers who promote these products to teenagers. This more targeted approach could make implementation and enforcement more feasible and reduce the burden on implementing authorities and regulated entities.”
Kelly Bonner, Associate Attorney at Duane Morris LLP
“We expected the bill to fail,” Bonner said, “but given the increased attention on personal care products and their health effects, and the ongoing media attention on the issue, we would not be surprised to see similar bills introduced in other states that are active in regulating cosmetic ingredients, such as Washington and Oregon.”
“And once again, it’s important to remember that the issue here isn’t the safety of the ingredients, but that the ingredients aren’t being used as intended. This is something that should be discussed with caregivers and health professionals, not the cashier at a beauty retailer.”