Recent research published in journals natural aging reported that small extracellular vesicles (sEVs) in the plasma of young mice interfere with pre-existing aging.

study: Small extracellular vesicles from young plasma reverse age-related functional decline by improving mitochondrial energy metabolism. Image credit: Ground Picture / Shutterstock.com
Can aging be reversed?
Recent studies have demonstrated that blood from young mice can connect the circulatory system through metachronous parabiosis and rejuvenate the brain, liver, bones, skeletal muscle, pancreas, and heart of older mice. Similarly, plasma infusion can reproduce the phenotype conferred by blood exchange in metachronous parabiosis.
Intensive efforts to identify plasma factors involved in reversing age-related functional impairments have led to the discovery of rejuvenating, rejuvenating, or anti-aging factors. However, their mechanism of action remains unclear.
About research
EVs are nanosized membrane vesicles that circulate in the bloodstream and function as intercellular messengers by exchanging cargo between cells. In the current study, researchers investigated the rejuvenating effects of sEVs.
The researchers first purified sEVs from young and old male mice, aged 2 and 20 months, respectively. Next, it was assessed whether young plasma sEVs could extend the lifespan of old mice. Old mice received an equal volume of young plasma sEV or phosphate-buffered saline (PBS) intravenously once a week until death.
research result
sEV administration to young mice significantly extended the frailty index score and median lifespan of old mice by 12.4%. Furthermore, the estimated biological age of the sEV-treated mice was 24 months, whereas their chronological age was 15.1 months.
Treatment with young sEVs improved testosterone levels, sperm count, motility, sperm chromatin integrity, and decreased sperm DNA fragmentation. Fertile female mice were mated with young or old mice, and embryo implantation was visually inspected after 4.5 days.
Mating with young males resulted in 7.7 implantation sites per pregnant female, whereas no implantation sites could be established in older mice. Nevertheless, treatment of aged mice with young sEVs significantly ameliorated fertility defects at 3.4 sites per pregnant female.
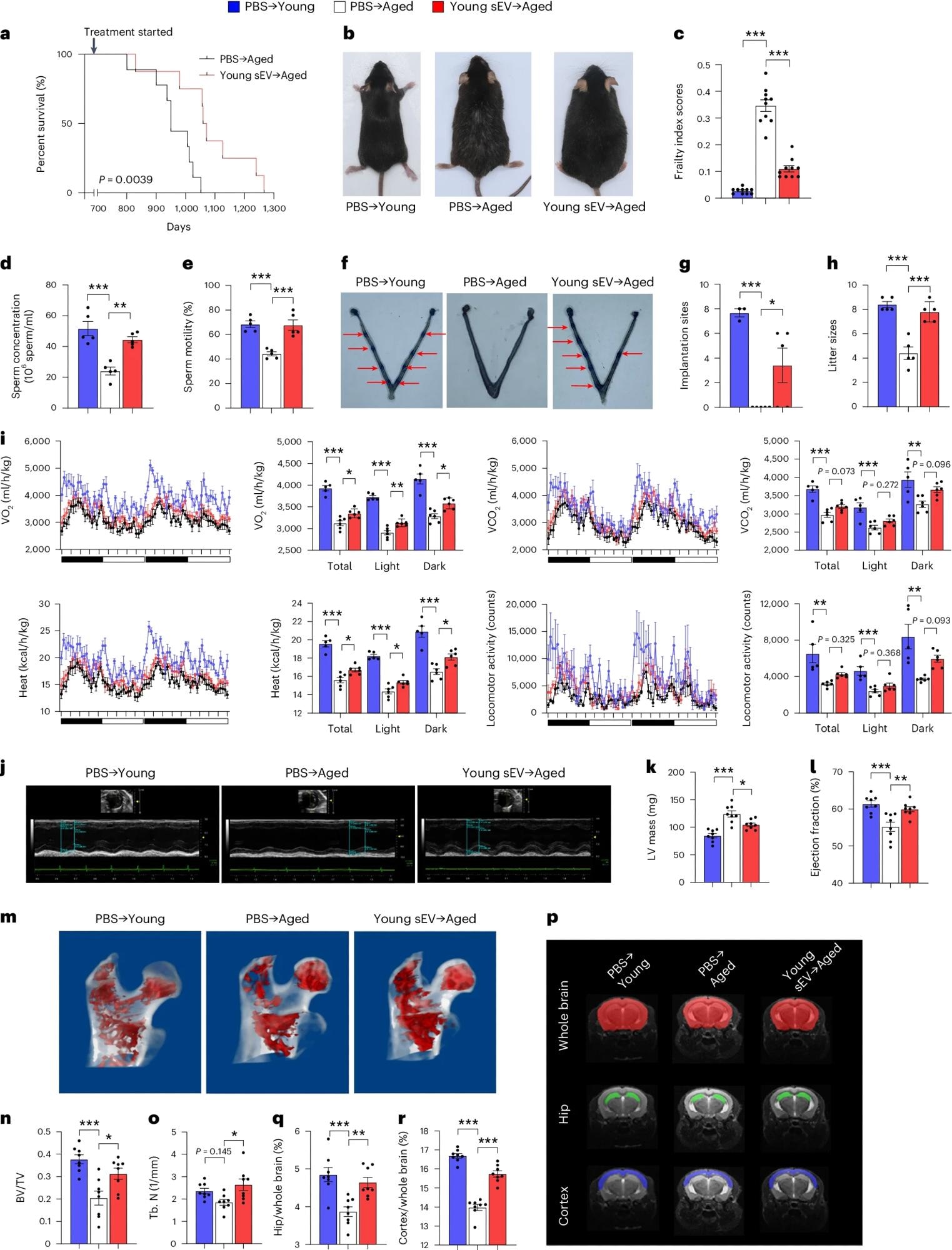 Old male mice (20 months old) were injected intravenously once a week with 200 μl of PBS or young sEVs (2 months old) and monitored to determine survival or systemic physiology. As a control group, young male mice (2 months old) were simultaneously injected with PBS. beKaplan-Meier survival curves for each group (n = 8–9). bRepresentative images of mice in each group after 7 months of treatment. cmean frailty index score for each group after 4 months of treatment (n = 10). d,esperm count and motility in each group (n = 5). f,g, Number of implantation sites visible as blue bands in the uterus of female mice mated with male mice of each group. Representative images (red arrows indicate implantation sites) and quantitative data (PBS → n = 3 for young, n = 5 for others) are shown. hthe number of pups born by male mice in each group (n = 5). IIndirect calorimetry of O2 consumption, CO2 Release, thermogenesis, and locomotor activity for each group (PBS → n = 5 for young, n = 6 for others). j–I, echocardiographic measurements of cardiac dimensions and indicators of cardiac function in each group. Representative M-mode echocardiograms and quantitative values of LV mass and EF (n = 8) are shown. meter–ahMicro-CT analysis of the trabecular microstructure of the proximal femur in each group. Representative 3D images of the proximal femur and quantitative values of BV/TV and Tb.N (n = 8) are shown. p–r, MRI-based morphometric analysis of the hippocampus and cortex of each group. Representative his MRI scans of slices are shown and volume ratios (hippocampus/whole brain and cortex/whole brain) were calculated (n = 8). Significance was determined using the log-rank test. be Use one-way analysis of variance followed by Dunnett’s multiple comparisons test. c–e, g–I, k, I, n, ah, q and r. *P < 0.05, **P < 0.01, ***P < 0.005.
Old male mice (20 months old) were injected intravenously once a week with 200 μl of PBS or young sEVs (2 months old) and monitored to determine survival or systemic physiology. As a control group, young male mice (2 months old) were simultaneously injected with PBS. beKaplan-Meier survival curves for each group (n = 8–9). bRepresentative images of mice in each group after 7 months of treatment. cmean frailty index score for each group after 4 months of treatment (n = 10). d,esperm count and motility in each group (n = 5). f,g, Number of implantation sites visible as blue bands in the uterus of female mice mated with male mice of each group. Representative images (red arrows indicate implantation sites) and quantitative data (PBS → n = 3 for young, n = 5 for others) are shown. hthe number of pups born by male mice in each group (n = 5). IIndirect calorimetry of O2 consumption, CO2 Release, thermogenesis, and locomotor activity for each group (PBS → n = 5 for young, n = 6 for others). j–I, echocardiographic measurements of cardiac dimensions and indicators of cardiac function in each group. Representative M-mode echocardiograms and quantitative values of LV mass and EF (n = 8) are shown. meter–ahMicro-CT analysis of the trabecular microstructure of the proximal femur in each group. Representative 3D images of the proximal femur and quantitative values of BV/TV and Tb.N (n = 8) are shown. p–r, MRI-based morphometric analysis of the hippocampus and cortex of each group. Representative his MRI scans of slices are shown and volume ratios (hippocampus/whole brain and cortex/whole brain) were calculated (n = 8). Significance was determined using the log-rank test. be Use one-way analysis of variance followed by Dunnett’s multiple comparisons test. c–e, g–I, k, I, n, ah, q and r. *P < 0.05, **P < 0.01, ***P < 0.005.
After one month of continuous mating, young and old males produced eight and four offspring per litter, respectively. Furthermore, when older mice were treated with young sEVs, they produced approximately the same number of pups as young males.
Older mice treated with young sEVs also showed higher oxygen consumption and carbon dioxide production than PBS recipients, thus suggesting that metabolic health was partially restored to the levels of young mice. Treatment also significantly improved echocardiographic parameters and bone structure in the aged mice. Magnetic resonance imaging (MRI) showed cortical atrophy in aged mice, which was alleviated by treatment with young sEVs.
Older mice showed increased senescence-associated β-galactosidase (SA-β-gal) activity in the spleen, kidney, liver, lung, testis, and hippocampus compared to young mice. However, when old mice were treated with young sEVs, even for 2 weeks, SA-β-gal levels in these organs rapidly decreased.
Young sEV treatment also restored intracellular reactive oxygen species (ROS) levels in old mice to those observed in young mice and removed excess glycation end product accumulation. Proteomic analysis showed that young sEVs could reverse age-related degenerative changes and exert rejuvenating effects.
The researchers also evaluated the effects of treatment with sEVs isolated from young humans on age-related functional impairments in mice. To this end, treating aged mice with sEVs derived from young humans effectively ameliorated cognitive impairment, enhanced endurance capacity, and supported the recovery of mitochondrial activity in muscle.
Although sEVs efficiently transport nucleic acids, proteins, and lipids, most studies have focused on the ribonucleic acid (RNA) components of sEVs, especially microRNAs (miRNAs). To elucidate the role of specific miRNAs in plasma sEVs, we evaluated the differential expression of miRNAs in plasma between young and aged mice through small RNA deep sequencing.
Additional experiments identified representative miRNA cargoes for young and aged sEVs. More specifically, miR-455-3p, miR-144-3p, and miR-149-5p represent the young state, and miR-34a-5p, miR-29a-3p, and miR-29c-3p represent the aging state. It represents the state of In particular, peroxisome proliferator-activated receptor gamma coactivator 1 alpha (PGC-1α) is a common target negatively regulated by miR-34a-5p, miR-29a-3p, and miR-29c-3p. was.
Furthermore, miR-455-3p, miR-144-3p, and miR-149-5p may be indirect stimulators of PGC-1α, as their downstream target genes show an inverse correlation with PGC-1α. There is a gender. Aged mice showed significantly reduced PGC-1α expression levels in muscle and hippocampus compared to young mice, and young sEV treatment of aged mice increased its expression.
Silencing PGC-1α using small interfering RNA (siRNA) significantly reduced the benefits on mitochondrial respiration observed with young sEV treatment. Additional studies showed that miR-455-3p, miR-144-3p, and miR-149-5p are rejuvenating miRNAs in young sEVs, whereas miR-34a-5p, miR-29a-3p, and miR -29c-3p was confirmed to be a rejuvenating miRNA. sEV was an aging-promoting miRNA.
conclusion
Repeated administration of young sEVs improved physiological function and physical performance in aged mice. In the short term, young sEV treatment led to rapid systemic improvement. In contrast, young sEV treatment restored the performance and properties of aged tissues to the levels observed in young mice over an extended period of time.
Taken together, the findings indicate that young sEVs may lead to promising opportunities to rejuvenate aging tissues and improve well-being and longevity.
Reference magazines:
- Chen, X., Luo, Y., Zhu, Q. other. (2024). Small extracellular vesicles from young plasma reverse age-related functional decline by improving mitochondrial energy metabolism. natural aging. doi:10.1038/s43587-024-00612-4

